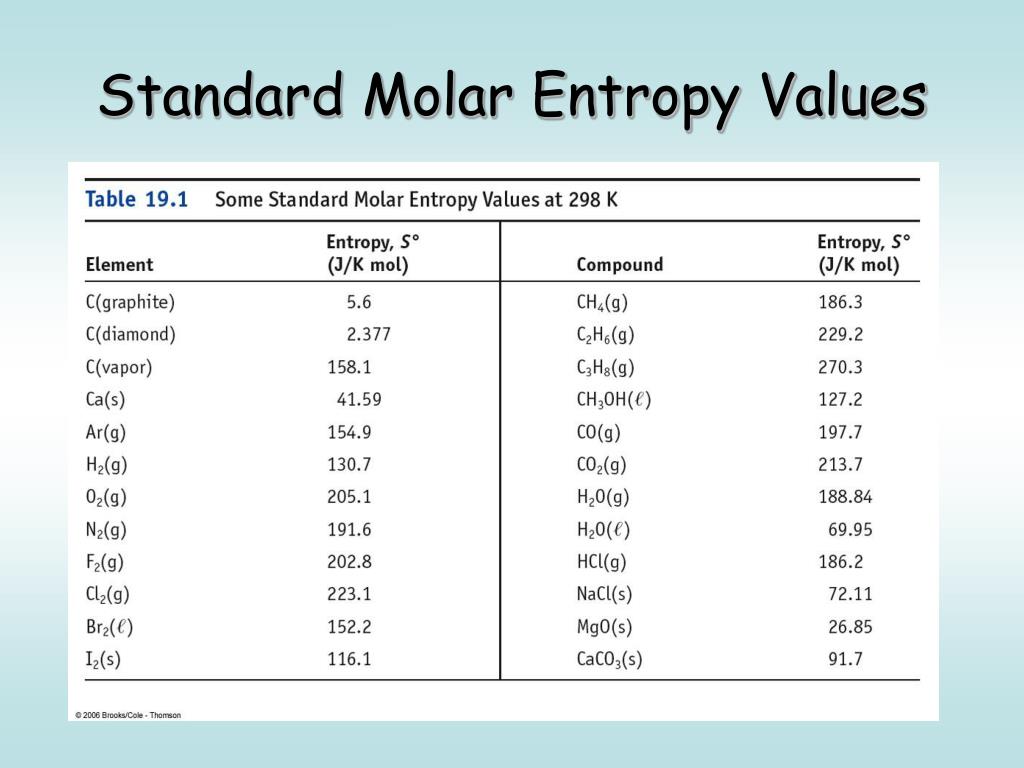
Miller, Computational Methods of Neutron Transport, American Nuclear Society, 1993, ISBN: 2-4. Hetrick, Dynamics of Nuclear Reactors, American Nuclear Society, 1993, ISBN: 3-2. Neuhold, Introductory Nuclear Reactor Dynamics, American Nuclear Society, 1985, ISBN: 9-4. Units of entropy As entropy change is expressed as heat divided by temperature, the units of entropy are calories per degree (expressed as entropy unit eu. Bezella, Introductory Nuclear Reactor Statics, American Nuclear Society, Revised edition (1989), 1989, ISBN: 3-2. Department of Energy, Nuclear Physics and Reactor Theory. DOE Fundamentals Handbook, Volume 1 and 2. January 1993. Robert Reed Burn, Introduction to Nuclear Reactor Operation, 1988.Physics of Nuclear Kinetics. Addison-Wesley Pub. Nuclear and Particle Physics. Clarendon Press 1 edition, 1991, ISBN: 978-0198520467 Nuclear Reactor Engineering: Reactor Systems Engineering, Springer 4th edition, 1994, ISBN: 978-0412985317 Stacey, Nuclear Reactor Physics, John Wiley & Sons, 2001, ISBN: 0- 471-39127-1. Baratta, Introduction to Nuclear Engineering, 3d ed., Prentice-Hall, 2001, ISBN: 8-1. Lamarsh, Introduction to Nuclear Reactor Theory, 2nd ed., Addison-Wesley, Reading, MA (1983). The fact that the absolute value of specific entropy is unknown is not a problem, however, because it is the change in specific entropy (∆s) and not the absolute value that is important in practical problems. For example, the specific entropy of water or steam is given using the reference that the specific entropy of water is zero at 0.01☌ and normal atmospheric pressure, where s = 0.00 kJ/kg. Normally, the entropy of a substance is given with respect to some reference value. In general, specific entropy is a property of a substance, like pressure, temperature, and volume, but it cannot be measured directly. where p is the pressure and V is the volume of the gas. unit of thermodynamic entropy, usually denoted 'e.u.' and equal to one calorie per kelvin per mole, or 4.184 joules per kelvin per mole. 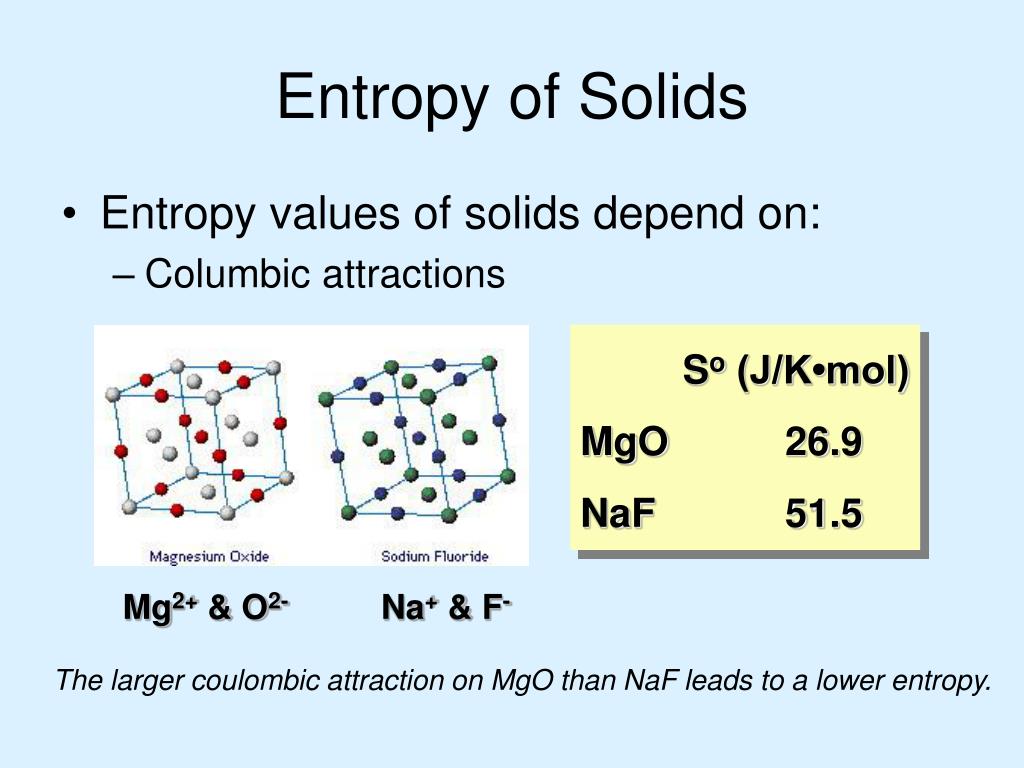
Substituting for the definition of work for a gas. where E is the internal energy and W is the work done by the system. We begin by using the first law of thermodynamics: dE dQ - dW. Because entropy tells so much about the usefulness of an amount of heat transferred in performing work, the steam tables include values of specific entropy (s = S/m) as part of the information tabulated. For gases, there are two possible ways to evaluate the change in entropy. M = mass (kg) T-s diagram of Rankine CycleĮntropy quantifies the energy of a substance that is no longer available to perform useful work. Other units in the category of Entropy include Joule Per Kelvin (J/K). It equals to the total entropy (S) divided by the total mass (m). 
The specific entropy (s) of a substance is its entropy per unit mass. Engineers use the specific entropy in thermodynamic analysis more than the entropy itself. and equal to one calorie per kelvin per mole, or 4.184 joules per. The entropy can be made into an intensive, or specific, variable by dividing by the mass. unit of thermodynamic entropy, usually denoted e.u.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
